PLOS Biology Paper Wins Omenn Prize for Viral Evasion Story
We talk to the authors of a PLOS Biology research article published last May that has just won the Omenn Prize for the best article published in 2013. The Omenn Prize is awarded annually by the Evolution, Medicine, & Public Health Foundation to authors of articles related to “evolution in the context of medicine and public health,” and the winner was picked from a tough long-list of 47 papers. Four other papers, including two from our sister journal PLOS Pathogens, were cited for “honorable mention” (Graves et al. Huijben et al.).
The PLOS Biology paper looks at how an essential mammalian protein – the transferrin receptor, TfR1 – evolves in the face of contrasting selective pressures. TfR1 is a protein that sits on the membrane of our cells and mediates the regulated uptake of iron. TfR1 is stuck in the horns of a dilemma. On the one hand, it has to be able to bind its functional partners – the iron-loaded plasma protein transferrin, and a negative regulator protein called HFE; this requirement constrains the sequence and structure of TfR1 through evolutionary time. On the other hand, it has to evade viruses that exploit its handy cell-surface location, such as arenaviruses and the rodent retrovirus MMTV.
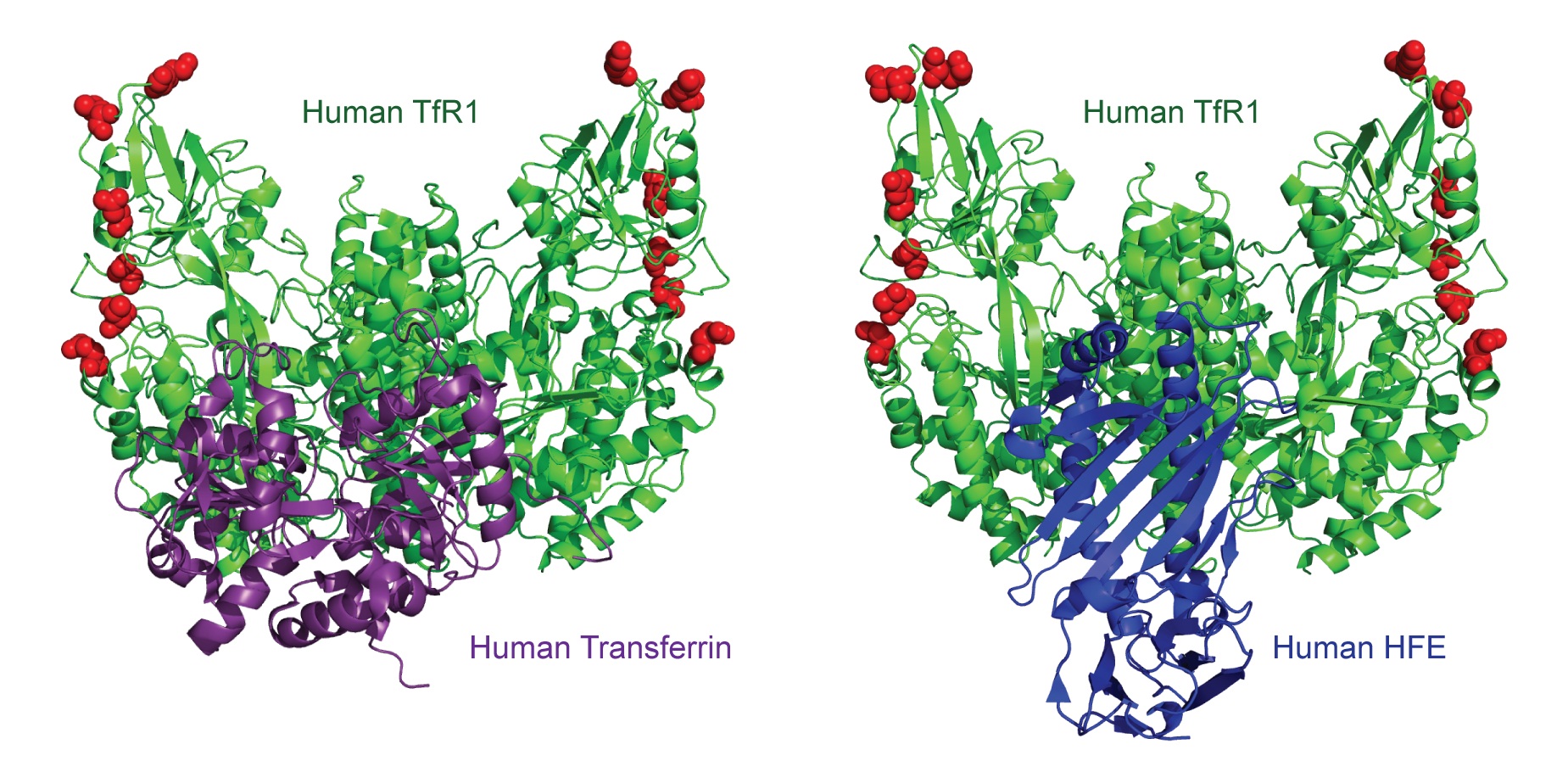
The authors compared sequences of TfR1 from various mammalian hosts and then expressed them on the surface of cells to check a) their ability to confer vulnerability or resistance to MMTV and arenaviruses such as Machupo, Junin and Guanarito virus and b) their ability to bind to transferrin. This image from the paper summarises the central finding – how TfR1 (green) manages to square this circle by evolving rapidly (red) to change the outer surfaces that are hijacked by viruses while keeping constant the central surfaces that it uses to bind transferrin and HFE (purple, blue).
First author Ann Demogines and lead author Sara Sawyer – both from the University of Texas at Austin – told us how the study first arose and then evolved into the paper that you can now read on our website.
Sawyer recalls the exact point at which the project started: “In the first year of my faculty position, Welkin Johnson invited me to give a talk at the New England Primate Research Center. While I was there, I had a 45 minute meeting with his colleague, Mike Farzan [also a co-author]. Mike had just discovered TfR1 as the cellular receptor for arenaviruses, and suggested to me that this might be a molecule that is engaged in an evolutionary arms race. While ideas like this often arise out of conversations between scientists, I remember having a gut reaction that this was something worth pursuing.”

Demogines, who received $5000 from the Foundation, describes how the spectacular arrangement of the evolutionarily selected sites emerged: “I am still amazed to this day by the results of the evolutionary analysis. We were able to take DNA sequence from just 7 species and computationally predict six sites under selection. These sites were scattered on the linear diagram of the protein, and didn’t make much sense to us. But, when we placed them onto the 3D crystal structure they formed a beautiful ridge going straight down the outer surface of the receptor. That was a great day in the lab. We knew this had to mean something!”
Demogines goes on to think about the implications of her paper and related studies: “This work really gets me excited about the future of evolutionary analysis applied in biomedical research. As we collect the genome sequences from more and more species, especially rodents and bats which are major reservoirs for zoonotic and potentially zoonotic viruses, we should be able to do this type of analysis more and more. This type of analysis has many applications: allowing us to identify critical cofactors involved in the viral lifecycle, viral binding sites, and potentially novel drug targets. It can also be used to study interactions with bacterial pathogens, although this has not yet been extensively explored.”
If you’d like to find out more about this elegant study, why not read the article itself, or the accompanying Primer written by John Coffin:
“Dual Host-Virus Arms Races Shape an Essential Housekeeping Protein” by Ann Demogines, Jonathan Abraham, Hyeryun Choe, Michael Farzan and Sara L. Sawyer. DOI: 10.1371/journal.pbio.1001571
“Virions at the Gates: Receptors and the Host–Virus Arms Race” by John M. Coffin. DOI: 10.1371/journal.pbio.1001574
