This week in PLOS Biology
In PLOS Biology this week, you can read about a new mechanism for incomplete puberty and infertility, and the control of embryonic tissue separation by ephrin/Eph pairs.
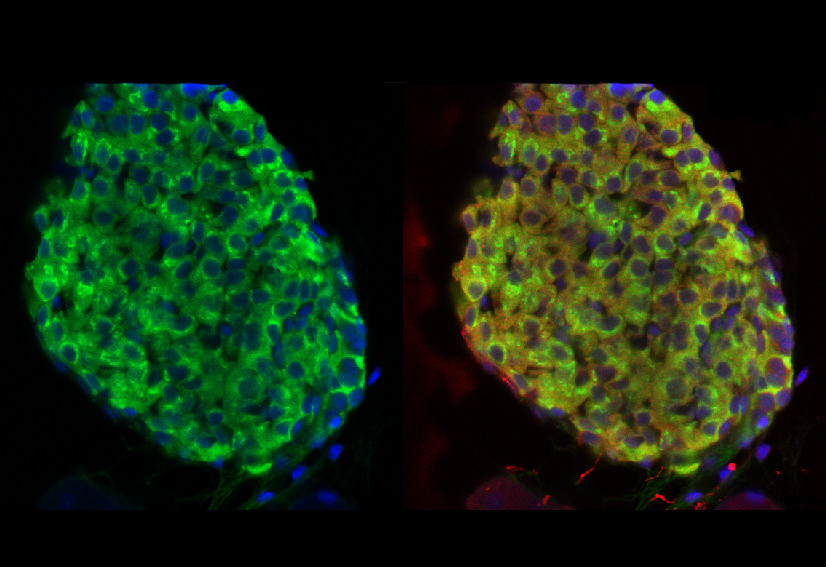
Rab3α Scaffolding and the Control of Puberty
Brooke Tata, Lukas Huijbregts, Nicolas de Roux and colleagues have described a new genetic syndrome, observed in three brothers, involving a complex set of symptoms such as incomplete puberty and non-autoimmune diabetes. Genetic analyses of the family revealed that they suffered from deletion of 15 nucelotides from the DMXL2 gene. This gene encodes Rabconnectin-3α, a protein involved in scaffolding the regulators of small GTPase Rab3α intracellular trafficking (see also this recent PLOS Biology paper). The authors found involvement for the protein in both GnRH secretion in the brain and insulin secretion in the pancreas – providing clues as to how decreased DMXL2 could be responsible for incomplete puberty and diabetes in this syndrome. Read more in the accompanying synopsis.
Embryonic Tissue Separation Depends on Specific Combinations of Ligand and Receptor Pairs
How embryonic tissues separate from each other to shape the developing organism is a fundamental question in developmental biology. In vertebrates, this process relies on local repulsive reactions specifically generated at contacts between cells of different types. We believe from recent evidence that the interaction is between surface proteins called ephrins and Eph receptors on the opposing groups of cells. However it’s been unclear how this repulsion is restricted to the tissue boundary and doesn’t take place within the tissues themselves. A new paper by Nazanin Rohani, Francois Fagotto and colleagues used a Xenopus model, and found that the concentration and binding affinities of specific ligand–receptor pairs largely explain the specificity of this key developmental program. Read more in the accompanying synopsis.