Understanding Images: Plants Limit Crossovers
Authors: Javier Varas and Mónica Pradillo from Universidad Complutense de Madrid, Madrid, Spain.
Competing interests: Javier Varas and Mónica Pradillo are authors of the work discussed in this blog.
In the July issue of PLOS Genetics, the regulation of meiotic crossover in plants was investigated by Varas et al.
Meiosis is a key event in the life of all sexually reproductive organisms, with major implications on the transmission of genetic information and on genome evolution. This process requires specialized features to generate haploid gametes from diploid cells. Meiosis consists of two divisions without an intervening S phase. During the first meiotic division, the formation of chiasmata between homologous chromosomes holds them together to promote their accurate segregation. These connections prevent uneven distribution of chromosomes that could lead to aneuploidy, as in Down syndrome. Chiasmata are the cytological manifestations of reciprocal interchanges of DNA (crossovers) and homologous recombination is the molecular process underlying the formation of crossovers.

Meiotic crossovers result from the repair of double-strand DNA breaks induced by SPO11. These double-strand breaks can be processed by multiple recombination pathways with specific intermediates to generate either non-reciprocal interchanges, non-crossovers, or reciprocal interchanges, crossovers. Crossover distribution is non-random and is tightly regulated by several mechanisms. All chromosomes receive at least one crossover (an obligate crossover), and formation of a crossover at a given site generally reduces crossover formation at adjacent regions – a phenomenon known as positive interference. In addition, crossover homeostasis ensures a stable number of crossovers despite variability in the number of double-strand breaks.
Meiotic Recombination in Plants
In plants, most of our knowledge about meiotic recombination has come through studies of the model species Arabidopsis thaliana. We have selected this species to demonstrate that in plants, as in yeast and mouse, increased double-strand break formation is not accompanied by increased crossover formation. Our study describes the meiotic phenotype of the Atfas1-4 mutant, defective for the large subunit of the histone chaperone CAF-1. This mutant shows developmental abnormalities; their stems, roots, siliques (seed pods) and flowers are reduced in size and the leaves are serrated. It also has reduced heterochromatin content and increased frequency of somatic homologous recombination.
Regulation of Crossover Formation in Arabidopsis

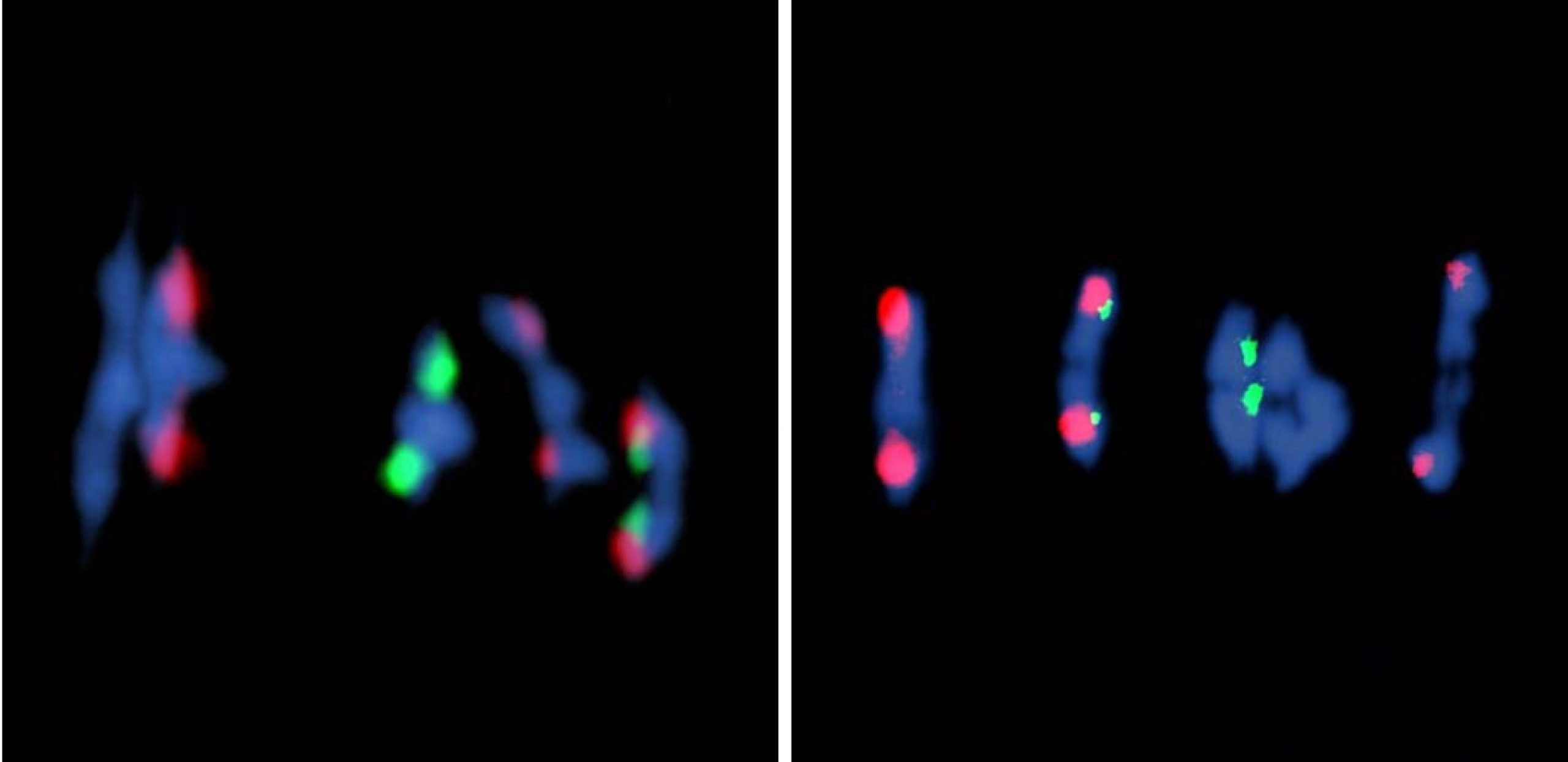
Arabidopsis has approximately 15 times more double-strand breaks than crossovers. To estimate the number of double-strand breaks in Atfas1-4, we counted foci corresponding to phosphorylated histone H2AX (γH2AX), a sensitive marker that can be used to examine the DNA damage produced and the subsequent repair of the DNA lesion, and the recombinases AtRAD51 and AtDMC1 which are involved in DNA strand invasion during the beginning of the homologous recombination process. In our issue image, the cell on the right corresponds to a dual immuno-localization of the axial element protein AtASY1 (shown in green in the above image) and AtRAD51 (shown in red). We concluded that the number of double-strand breaks is increased by more than 50% in Atfas1-4.
These extra double-strand breaks could be processed by the different pathways which participate in normal conditions; nevertheless, we did not detect an increase in crossovers. We tested whether non-crossover gene conversion frequencies change in Atfas1-4 relative to wild-type plants. To estimate the gene conversion frequencies we used lines with fluorescent pollen grains. Pollen tetrads from plants heterozygous for fluorescent and non-fluorescent alleles present two fluorescent pollen grains and two non-fluorescent pollen grains (top left tetrad in the figure). If gene conversion occurs, a non-Mendelian 3:1 ratio is observed (bottom left tetrad). In the mutant the frequency of these tetrads was increased compared to the wild-type. Therefore, in Atfas1-4 the excess of double-strand breaks produces an increase in the frequency of gene conversion events.
Implications of Findings
Our findings show that the number of crossovers can be constrained in plant species even when the number of double-strand breaks increases during meiosis. The extra double strand breaks produced in Atfas1-4 are processed to non-crossovers in order to keep the same crossover frequency as the wild type, resulting in an increase in gene conversion frequency. These results highlight the complex regulation of crossover formation during Arabidopsis meiosis. Our results demonstrate that despite considerable divergence in molecular components that drive meiosis, there is conservation of the homeostatic mechanism which regulates recombination from fungi to plants to animals.
Original Article
Varas J, Sánchez-Morán E, Copenhaver GP, Santos JL, Pradillo M (2015) Analysis of the Relationships between DNA Double-Strand Breaks, Synaptonemal Complex and Crossovers Using the Atfas1-4 PLoS Genet 11(7): e1005301. doi:10.1371/journal.pgen.1005301
