Accidents Will Happen… Introducing LabCIRS
Ever wondered how a laboratory (or a scientific research department) should best manage its day-to-day operations? In a recently published PLOS Biology Community Page article, our Editorial Board member Ulrich Dirnagl and colleagues outline a simple system to manage errors in the lab and foster a mature ‘error culture.’
Critical incident reporting isn’t a new idea. It’s been used in clinical medicine since 1978 and is internationally recognized as a valuable tool for improving patient safety. Dr Dirnagl and colleagues now adapt and refine this tool to fit a modern (pre-clinical) biomedical research setting.
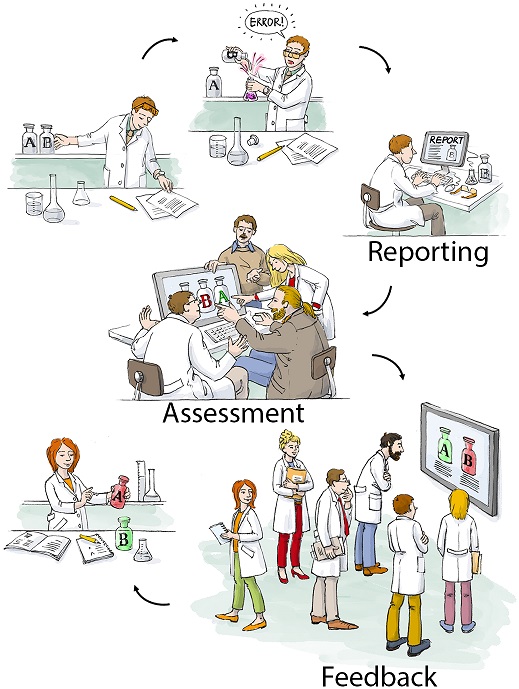
The lab critical incident reporting system (LabCIRS) was been implemented and tested in-house within the authors’ own multi-disciplinary Department of Experimental Neurology at the Charité Universitätsmedizin Berlin (a demo version is available here). In a key feature, LabCIRS allows for anonymous reporting of errors and incidents, from incorrect labeling of cuvettes to accidental mouse escapes. Reports are analyzed by experts, and results fed back to prevent recurrence.
The authors find that communication and management of errors improved after implementing LabCIRS and say “We are convinced that LabCIRS has clearly improved the quality of our work and made the laboratory a safer and more communicative environment.” Importantly, the open-source software underlying LabCIRs is now made freely available along with the article. The hope is that others in the scientific community will adapt and implement similar systems to help improve research quality control.
Featured image credit: Flickr user horiavarian
