The XV Collection: Perverse Outcomes of Novel Therapies
by Andrew Read
Yale professor Steve Stearns once warned that the transition from Young Turk to Old Turkey happens quickly. He was right. Being an Old Turkey has challenges, not least that you more readily spot ignorance but you have less bandwidth to think it through. So, it is a total joy to come across an analysis you wish you’d had time to do yourself. The paper I have chosen for the PLOS Biology XV Collection is one of those. In a 2014 Essay, three Young Turks — Pedro Vale, Andy Fenton and Sam Brown — considered a strategy said by others to be a solution to one of the great health challenges of the 21st century. Before their paper, all I could see was a jumble of plusses and minuses. They sorted that out.
Prompted by the antimicrobial resistance crisis – said by some to be a bigger threat to humanity than terrorism – the search is on for drugs that can treat patients infected with resistant bugs – ideally without causing resistance themselves. One idea is to find drugs which make bugs less lethal (for example, by inactivating secreted bacterial toxins) or by making the patient more robust (for example, by enhancing tissue repair). The thinking is that such drugs would make the patient feel better and – because the bug is allowed to survive – resistance evolution won’t ensue.
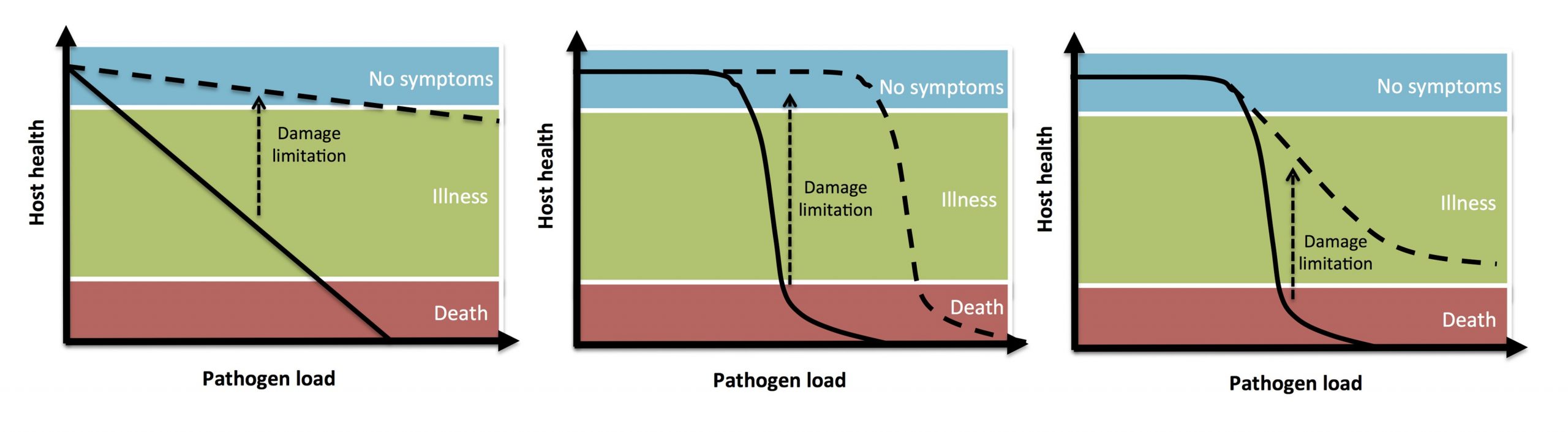
The claim that these ‘damage-limitation’ drugs would be evolution-proof set off alarm bells in my head when I first heard it soon after the turn of the century. The only thing that stops evolution is death. Otherwise, as actor Jeff Goldblum put it in Jurassic Park “Life, uh, finds a way.” Vale et al. provide what I consider the first sensible analysis of the epidemiological and evolution consequences of damage-limiting drugs. In a few pithy paragraphs (and seven lines of algebra), they show there are many possible consequences, not all of which are good. Most immediate are the transmission consequences. Not only will bugs be left alive to transmit, but infectious people will be harder to spot and, being less sick, more likely to be in contact with susceptible people. And the evolutionary outcomes also need not be good. For instance, if toxin production gives pathogens fitness advantages, as it almost always does, inactivating toxins with drugs might select for bugs that produce more toxin than the drugs can deal with – or bugs that can produce other toxins. None of those potentially harmful outcomes have to eventuate, as Vale et al. make clear, but they very well might.
The risk of harm is not an argument against developing damage-limiting drugs. But it is a strong argument for not considering them as magic bullets. Just like conventional drugs, their impact on pathogen transmission and evolution need to be studied at all stages of the discovery pipeline – and post-roll out. Vale et al. provide the roadmap for things to look for. It’s a paper I wish I’d written.
Vale PF, Fenton A, Brown SP (2014) Limiting Damage during Infection: Lessons from Infection Tolerance for Novel Therapeutics. PLoS Biol 12(1): e1001769. https://doi.org/10.1371/journal.pbio.1001769
Andrew Read is an Evan Pugh University Professor of Biology and Entomology at The Pennsylvania State University, and is a member of the PLOS Biology Editorial Board.
This blog post is the eighth in a series of twelve, forming PLOS Biology’s XV Collection, celebrating 15 years of outstanding open science; read Lauren Richardson’s blog for more information.
Featured image credit: Flickr use: NIAID
Andrew Read image credit: Brad Ziegler

