This week in PLOS Biology
In PLOS Biology this week you can read about wolves in Yellowstone National Park, regulation of hair growth, how proteins evolve new RNA-binding functions and the regulation of heat-shock response by histone demethylation.
Yellowstone Wolves and their Effects on Natural Systems

Wolves were re-introduced to Yellowstone National Park in 1995 and 1996 after a 70-year absence. Since then, wolves have had effects on Yellowstone that ripple across the entire structure of the food web that defines biodiversity in the Northern Rockies ecosystem. In a new essay, Andy Dobson discusses the different ecological interpretations of the effects wolves have had on the ecosystem. He argues that ecology in general needs more ecosystem-level studies of species interactions, which ultimately drive levels of biodiversity. A new generation of scientists who can grapple with the vast complexities of these questions is needed.
Hair Growth is Regulated by Macrophages
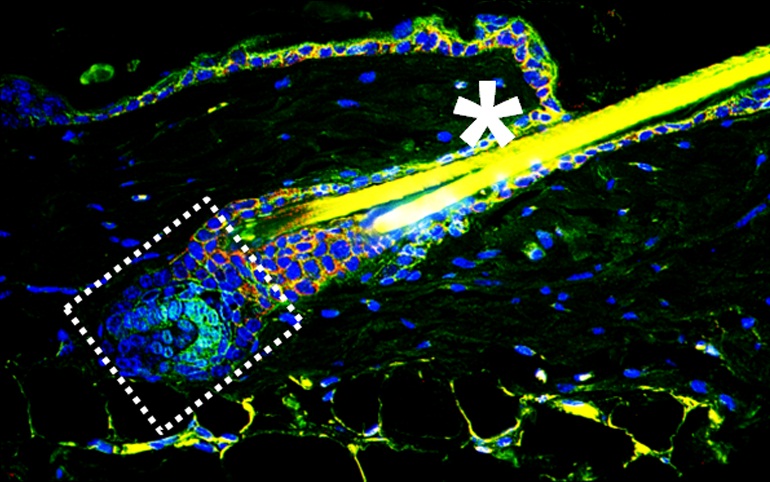
The cyclic life of hair follicles consists of recurring phases of growth, decay, and rest. Using a mouse model, Donatello Castellana, Ralk Paus & Mirna Perez-Moreno report that macrophages in the skin signal to skin stem cells via Wnt ligands in the resting phase of the follicle life cycle. This tale of niche cells modulating stem cell behaviour could be relevant for the development of technologies with potential applications in regeneration, aging, and cancer.
From tRNA Loading to mRNA Splicing Via Promiscuity

New research by Lilian Lamech, Anna Mallam & Alan Lambowitz addresses the broad question of how proteins evolve new RNA-binding functions. Specifically, the way in which a fungal mitochondrial tyrosyl-tRNA synthetase (mtTyrRS) has evolved to promote the splicing of group I introns. Using small-angle X-ray scattering (SAXS) and biochemical assays, the study supports the theory of pre-adaptive or constructive neutral evolution, and identify a previously unappreciated role for non-specific interactions in the evolution of RNA-binding proteins and other multi-subunit protein and ribonucleoprotein complexes.
Recruiting Histone Demethylase to Target Genes
The methylation state of the histone subunits of nucleosomes regulates gene transcription, but the delicate balance of counteracting methyl-transferases and demethylases is crucial for establishment and maintenance of appropriate methylation. This paper by Mo-bin Cheng, Yan Zhang, Ye Zhang, Yu-fei Shen & colleagues reports how specific phosphorylation of the histone demethylase KDM3A by the kinase MSK1 allows it to be recruited to promoters by Stat1 in response to heat shock, permitting the coordinated expression of many genes in human cells.
